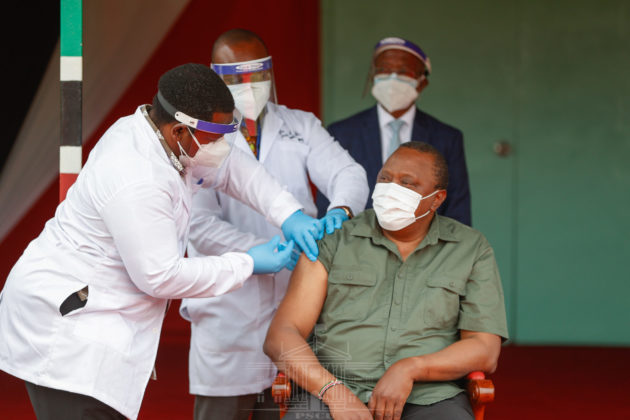
NAIROBI, Kenya, Apr 19 – A Science and Technology lobby group has emphasized the need to educate the public on the process of clinical trials on various drugs and vaccines like AstraZeneca which has been received with skepticism by several Kenyans.
The concerns arose following reports from countries, mainly in Europe, where some patients developed blood clots after getting the AstraZeneca jab. Deaths were also reported prompting some of the countries to suspend the jab.
According to the Alliance for Accelerating Excellence in Science in Africa, there is need to sensitize the public on the safety and effectiveness of the trials that were carried out in Kenya.
The organization’s Community Public Engagement Manager Lilian Mutengu said this will further boost their confidence in the use of various products.
“If people do not understand what is happening, whether a product is good and effective, it will not have real life impact because people will not appreciate it, people will not take it up,” she said during an interview with Capital News.
In October last year, Kenya joined the global efforts in search of an effective vaccine for COVID-19 with the start of a trial evaluating the AstraZeneca- the Oxford coronavirus vaccine.
The trial was hosted at KEMRI at its Kilifi-based KEMRI-Wellcome Trust Research Programme, under a longstanding collaboration between KEMRI, the University of Oxford and the Wellcome Trust in the UK.
“If people don’t understand the process, the issue of trust arise. What would be critical for the government and our leaders in the science world to come together and empower people to understand and appreciate the value science brings to the society,” she said.
In the Oxford trials, at least 40 frontline health CARE workers within Kilifi participated. Once the vaccine safety was confirmed, a further 360 volunteers were set to be recruited with possible expansion of the trial to Mombasa County.
She called on the government and other research organisations to priorities public engagement and sensitization to ensure the clinical trials are acceptable.
Mutengu said there is need for community and public management in such trials.
“If you have people understand the research process, right from the beginning to the end, not just because you need them to participate in clinical trials, but because you want to improve their research literacy,” she pointed out.
“That will go along way in ensuring science actually takes root in our society. A lot of economies have grown due to research and science. Without the community appreciating the value of science, the government will not priorities research department and clinical trials.”
It is such value that the developed world has adopted, she said, and was slowly metamorphosizing to vaccine nationalism.
“This is because they have appreciated the value of science. We need to do the same in Africa, we need to start funding research,” she said.
Further, she challenged the government to heighten disease surveillance and enhance regional integration in management of diseases like the COVID-19 pandemic.
For example, she said, there is need for harmonization of COVID-19 protocols within the region, to ensure people can seamlessly move without a lot of challenges.
“Why do we have these variations if we are a regional block?” she asked. “Some of these are the challenges that are clearly cut so that disease surveillance across the region.”
She also said there is a need for strengthening the regulatory framework for clinical trials across the region.