The board also announced that it would conduct a post-market surveillance within the next 45 days to weed out dodgy suppliers/FILE
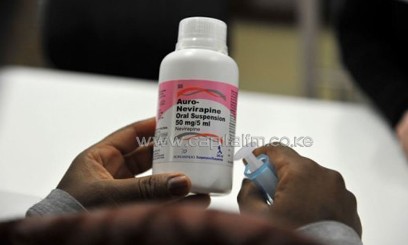
KMLTTB Chairman Abel Onyango told journalists on Monday that there were more than 100 unscrupulous suppliers of the kits because the board had only cleared 39 suppliers.
He explained that those who failed to meet the board’s ultimatum risked being jailed for five years, a Sh2 million fine or both if convicted.
“The board wishes to notify all institutions including government ones that within 45 days from today, anyone stocking or using HIV testing kits must ensure that they meet the KMLTTB thresholds,” he warned.
Onyango added that the medical board would work with the Kenya Revenue Authority to man the country’s entry points to ensure that no HIV testing kit made its way into the country without meeting the required standards.
The board also announced that it would conduct a post-market surveillance within the next 45 days to weed out dodgy suppliers.
“We are going to publish the names of the legally registered dealers so that even as you think of making any procurement relating to laboratory products then you know the recognised suppliers,” he said.
Concerns of fake HIV testing kits have formed debates among Kenyans but the government maintains that all is well.
Onyango however failed to respond to concerns surrounding the accuracy of the kits being supplied by the unscrupulous dealers.
“The public just needs to be aware of the correct government position. All the kits that will be used and distributed through the channels of the ministry will have gone through the critical elements for evaluation and validation,” he said.
Onyango added that the board was working with the Kenya Bureau of Statistics to get a mark of quality that would be stamped on all medical kits that met the required standards.
The board members also asked the government to consider setting up another medical laboratory to help in the clearance process.
“It is not enough that we only have one laboratory performing quality assurance for HIV testing. This country must move forward and establish regional centres of excellence to help us monitor the quality of all testing kits,” he explained.
This comes at a time when the government is already planning to import 10 million HIV testing kits from China and India.